 The operation of CURES complies with all applicable federal and state privacy and security laws and regulations. CURES is committed to the reduction of prescription drug abuse and diversion without affecting legitimate medical practice or patient care. The Department of Justice has also published a bulletin with information about the regulations, entitled, “ Adoption of Updated CURES Regulations.”Ĭontrolled Substance Utilization Review and Evaluation System California’s Prescription Drug Monitoring ProgramĬURES (Controlled Substance Utilization Review and Evaluation System) is a database of Schedule II, Schedule III, Schedule IV and Schedule V controlled substance prescriptions dispensed in California serving the public health, regulatory oversight agencies, and law enforcement. Relevant information concerning these regulations may be found on the following webpage of the Office of Attorney General’s website: The regulations revise and add to the CURES access and use regulations that took effect on July 1, 2020. Prescriptions and employee prescriptions must be reported.Updated regulations pertaining to the Controlled Substance Utilization Review and Evaluation System (CURES) became effective on August 15, 2022. Inpatient prescriptions dispensed are exempt from reporting. Prescriptions dispensed to assisted living facilities must be reported. Pharmacies dispensing to nursing homes are exempt from reporting. The following entities are exempt from reporting data:ĭispensing pursuant to a manufacturer’s indigent patient programĭispensing of covered substances by a practitioner of the healing arts to his patient in a bona fide medical emergency or when pharmaceutical services are not available.ĭispensing within an appropriately licensed narcotic maintenance treatment programĭispensing to inpatients in hospitals or nursing homes (exemption does not apply to assisted living)ĭispensing to inpatients in hospices (exemption does not apply to home hospice or hospice in an assisted living facility)ĭispensing of covered substances by veterinarians to animals within the usual course of their professional practice for a course of treatment to last seven days or less These elements and others of ASAP 4.2 please see Appendix A – ASAP 4.2 Specifications. The following elements are required by law to be reported. Virginia PMP is the state oversight agency andĪppriss will be acting as an agent of Virginia PMP in the collection of this information. Such reporting without individual authorization by the patient is allowed under HIPAA,Ĥ5CFR § 164.512, paragraphs (a) and (d). Of concern-gabapentin are required to collect and report their dispensing information. To amendments, it is the responsibility of dispensers to be aware of such updates asĪll dispensers of Schedule II - V controlled substance prescriptions, naloxone, and drugs The laws and regulations for reporting to the Virginia PMP are continuously subjected Dispensations must be reported within 24 hours, or theĭispenser’s next business day of dispensing the prescription. PMP through the authorized data collection vendor, Appriss, Inc.Įffective Novemthe Virginia PMP will begin requiring pharmacies andĭispensers to report controlled substance dispensations to the Virginia PMP via the Substance dispensing activities must be reported on regular intervals to the Virginia Private contractor that will collect all data and manage the technical aspects of theĪll dispensers of controlled substances must meet the reporting requirements set forthīy state law in a secure methodology and format.  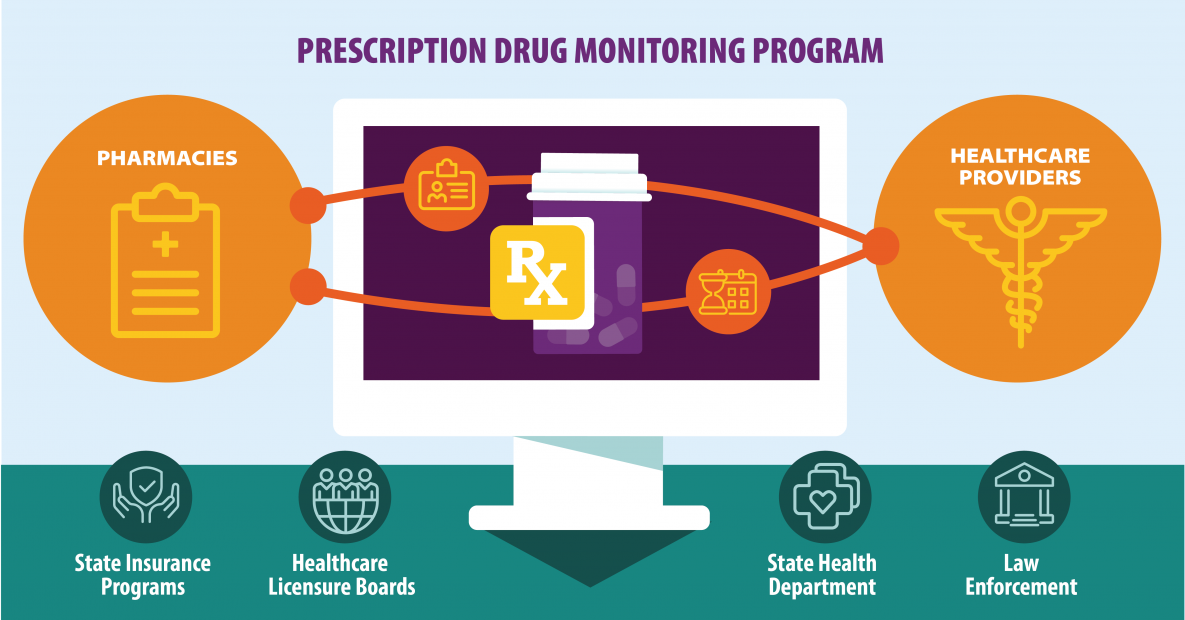
Report all controlled substances shipped to a patient that resides within theĭispensers will report the required dispensing information to Appriss, Inc. 
The programĪlso requires non-resident pharmacies licensed by the Virginia Board of Pharmacy to The program covers theĮntire state and requires all dispensers to report, within 24 hours, all prescriptionsĭispensed in Schedules II-V, naloxone, and drugs of concern-gabapentin. Of Health Professions (DHP) has established a program to monitor the prescribing andĭispensing of Schedule II, III, IV, and V controlled substances. In accordance with Code of Virginia §§ 54.1-2519 – 54.1-2525 the Virginia Department 43ġ Data Collection and Tracking Data Collection Overview 27ġ2 Appendix B – ASAP Zero Report Specifications. 26ġ1 Appendix A – ASAP 4.2 Specifications. 21ħ.2 Changing Passwords for Another User. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
